Attention-deficit hyperactivity disorder (ADHD) is a neurodevelopmental disorder characterised by the core symptoms hyperactivity, impulsivity and inattention. It is one of the most common psychiatric disorders, with worldwide prevalence estimates of 5.3% in children Reference Polanczyk, de Lima, Horta, Biederman and Rohde1 and 2.5% among adults. Reference Simon, Czobor, Balint, Meszaros and Bitter2 The disorder may cause substantial problems for the affected individuals and has a significant impact on society. Besides an important genetic factor, Reference Faraone, Perlis, Doyle, Smoller, Goralnick and Holmgren3 the aetiology of ADHD is largely unknown.
Vitamins are vital organic compounds that are primarily acquired through the diet, with the exception of vitamin D, which is synthesised when skin is exposed to sun. Vitamin deficiency may cause severe illness, because of the vitamins’ involvement in essential physiological and biochemical processes. For instance, vitamin A is important for growth and development, B vitamins are central in cell metabolism and nucleotide and neurotransmitter synthesis, vitamin D regulates cell and tissue growth and differentiation, and vitamin E plays a role in gene expression and in neurological function. Reference Bender4 As these processes are crucial for normal brain development and function, vitamin status and deficiencies may be important in ADHD. It has been reported that serum vitamin D levels are significantly lower in children and adolescents with ADHD compared with controls. Reference Bener, Kamal, Bener and Bhugra5–Reference Kamal, Bener and Ehlayel7 Accordingly, nutritional supplements including vitamins have repeatedly been suggested as treatment in ADHD. In two recent randomised controlled trials, it was found that ADHD symptoms were significantly reduced in adult ADHD patients who received a vitamin–mineral supplement Reference Rucklidge, Frampton, Gorman and Boggis8 and a vitamin B6-related drug, metadoxine, Reference Manor, Rubin, Daniely and Adler9 respectively. In another study, it was found that serum folate levels were significantly higher among adult ADHD patients than controls, whereas there was no statistically significant group differences for vitamin B12. Reference Karababa, Savas, Selek, Cicek, Cicek and Asoglu10
Owing to the important and neurologically relevant functions of vitamins and the lack of studies exploring this topic in ADHD, we measured serum levels of the major vitamin classes in a sample of adult ADHD patients and controls to determine whether vitamin levels are associated with ADHD diagnosis and psychiatric symptoms.
Method
Participants
Study participants were young adult individuals, who volunteered to participate in the project ‘ADHD: from clinical characterisation to molecular mechanisms’ at the University of Bergen. The majority of patients were enrolled in the study by responding to a postal invitation, as they were listed in a Norwegian national registry of adult ADHD patients. The remainder of the patients were recruited directly from out-patient clinics or psychiatrists, as described earlier. Reference Johansson, Halleland, Halmoy, Jacobsen, Landaas and Dramsdahl11 All patients had previously been diagnosed with ADHD according to the ICD-10 research protocol, 12 although allowing for the exceptions that the inattentive subtype in DSM-IV was sufficient for the diagnosis and that other psychiatric disorders could be present, as long as the ADHD criteria were fulfilled before the comorbid disorder appeared. Thus, the diagnostic criteria were very similar to the ADHD criteria in DSM-IV-TR. 13 During diagnostic workup, they were all considered as having childhood onset of the disorder, based on school reports and documentation obtained from patients, parents and other informants. Upon joining this study, 93% of all patients stated that they ‘believed that they had ADHD as children’. However, only 14% recalled that they had been treated for ADHD during childhood. This is in accordance with an earlier publication from our group, showing that a minority of adult ADHD patients recalled being diagnosed and treated during childhood. Reference Halmoy, Fasmer, Gillberg and Haavik14 There are multiple explanations for this discrepancy. Some patients could have received treatment during adolescence, although being diagnosed as children. For others, the severity of symptoms may not have been considered adequate to warrant diagnostic assessment or drug treatment, and for many, appropriate treatment was not available during their childhood. During the past decades, treatment guidelines for ADHD in Norway have changed several times, and the proportion of children, adolescents and adults receiving stimulant treatment has increased strongly. In addition, all retrospective information is subject to recall bias.
The control sample consisted of students at the University of Bergen, who volunteered to participate after having received oral information about the ADHD project in conjunction with lectures. In this study, participants aged between 18 and 40 who agreed to provide a blood sample were included. All participants were of Norwegian ancestry, and those who reported intellectual disability were excluded from the analyses. All participants signed a written informed consent, and the study was approved by the Norwegian Regional Medical Research Ethics Committee West (IRB #3 [FWA00009490, IRB00001872]).
Data collection
The participants filled in a questionnaire with 118 different items, including lifetime presence of psychiatric disorders and the standardised questionnaire the Adult ADHD Self-report Scale (ASRS). The ASRS is the World Health Organization's 18-item rating scale for measuring current symptoms of ADHD adapted to adults. Reference Kessler, Adler, Ames, Demler, Faraone and Hiripi15 It comprises two subscales, where symptoms of either inattention or hyperactivity and impulsivity are rated based on the DSM-IV-TR criteria for ADHD. 13 The participants indicated how often the symptoms occur on a Likert scale from ‘never’ to ‘very often’, giving a score between 0 and 4 points for each question. The questions on psychiatric morbidity other than ADHD, including alcohol misuse and problems with illicit drugs, were formulated, ‘Have you got or have you ever had….?’, and the alternative responses were ‘yes’ and ‘no’. The patients’ treating physicians provided a separate form confirming the ADHD diagnosis and with information regarding past and present treatment with stimulants Reference Hegvik, Jacobsen, Fredriksen, Zayats and Haavik16 and other psychopharmacological agents.
Venous blood samples were collected at the participants’ general practitioners (GPs) or by a research nurse at the Department of Biomedicine at the University of Bergen. Blood was withdrawn into serum gel tubes, which were left to coagulate before centrifugation and removal of serum. Serum samples collected by the GPs were shipped to Department of Biomedicine by postal mail (ambient temperature). The blood samples were visually inspected upon arrival, and samples that showed signs of haemolysis or degradation were excluded (n=2). The samples were stored at −80°C until they were thawed in 2013 and realiquoted for the biochemical analyses. The samples were collected in two time periods, 2004–2006 and 2010–2011, giving a total storage time of 2 to 9 years at −80°C. There was an equal representation of ADHD patients and controls at the two time points when samples were collected.
Biochemical analyses
The vitamins examined in this study were vitamin A (all-trans retinol), vitamin B2 (riboflavin), vitamin B3 (nicotinic acid), vitamin B6 (pyridoxal and pyridoxal 5’-phosphate), vitamin B9 (5-methyltetrahydrofolate), vitamin B12 (cobalamin and methylmalonic acid [MMA]), vitamin D (25-hydroxyvitamin D2 and 25-hydroxyvitamin D3) and vitamin E (α-tocopherol). Furthermore, cotinine levels were analysed to assess smoking status. All biomarkers were analysed in the laboratory of Bevital AS (www.bevital.no) by use of published methods. The concentrations of folate and cobalamin were determined using microbiological assays, Reference Kelleher and Broin17,Reference O'Broin and Kelleher18 whereas the remaining vitamins and metabolites, including cotinine, were analysed using liquid- or gas chromatography-tandem mass spectrometry. Reference Midttun, Hustad and Ueland19 Biomarkers that have been shown to be vulnerable to degradation during transport or storage were excluded. All biomarker analyses listed above were found to have sufficient quality to be included in the further analyses.
Statistical analyses
The statistical analyses were performed with the Statistical Package for Social Sciences versions 20 and 22 (SPSS, Inc., Chicago, IL, USA). The clinical characteristics of patients and controls, including their scores on the ASRS, were compared using chi-square and t-tests. To account for possible dephosphorylation of pyridoxal 5’-phosphate during sample transport, vitamin B6 levels were calculated as the total vitamin B6 aldehyde, that is, the sum of pyridoxal and pyridoxal phosphate. Reference Hustad, Eussen, Midttun, Ulvik, van de Kant and Morkrid20 The concentration of vitamin D was defined as the sum of 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3. Reference Dahl and Thorsby21 To evaluate vitamin B12 status, both cobalamin and MMA were analysed independently. The vitamin levels were not normally distributed and were therefore, and to avoid problems with outliers, analysed non-parametrically. Unadjusted levels of vitamins in patients and controls were analysed using Mann–Whitney U tests. Further analyses using binary logistic regression analyses adjusted for age and gender were performed to examine the influence of vitamin levels on ADHD and control status, with the levels categorised into quartiles based on the values in all participants together. Because alcoholism predisposes to vitamin B deficiency, Reference Cravo, Gloria, Selhub, Nadeau, Camilo and Resende22,Reference Savage and Lindenbaum23 the model was also adjusted for alcohol abuse in exploratory analyses. As the age mean and range were different in the patient and control groups, logistic regression analyses of vitamin levels and ADHD and control status including only the participants up to 30 years of age were additionally performed to investigate whether improved age-matching of the groups influenced the results. In addition to the logistic regression analyses, 10th percentile distributions of the vitamin levels were analysed using chi-square tests.
Because smoking is strongly associated with ADHD, cotinine levels were not included as a covariate in the regression analyses examining the relationship between the vitamins and ADHD and control status. To examine the possible effect of smoking on vitamin levels, we compared the median vitamin levels with Mann–Whitney U tests in ADHD patients categorised as non-smokers and smokers, as defined by having cotinine levels below or above 80 nmol/L, which is a widely used cut-off to distinguish smokers from non-smokers. Reference Jarvis, Tunstall-Pedoe, Feyerabend, Vesey and Saloojee24 We further conducted ANOVA analyses, also separating the heavy smokers who had cotinine levels of ≥1000 nmol/L from the moderate smokers, to explore a possible dose–response relationship between smoking and vitamin levels. The controls were not included in these analyses, since only a minority in this group was categorised as smokers.
As treatment with central stimulants was specific to the ADHD group and >90% of the patients reported ever being treated with stimulants, such treatment was difficult to examine as a factor independent from the ADHD diagnosis.
To evaluate the relationship between vitamin levels and ADHD symptoms, the total scores on the ASRS were used. The scores showed a near-normal distribution, and linear regression analyses were thus performed with untransformed values. Because the ASRS scores were higher among the patients, and generally were low in the controls, the analyses were also carried out in the ADHD patients separately. The analyses were performed with age and gender as covariates. However, as both the ASRS scores and the mean age were higher in the ADHD group, the analyses in all participants together were also performed without corrections for potential covariates, as they could attenuate putative associations. Additional analyses excluding the participants above 30 years of age were also performed, as were analyses of ASRS and vitamin levels with correction also for cotinine levels (non-, moderate and heavy smoking).
The level for a nominally statistically significant result was defined as a two-sided P-value of <0.05. The P-values are presented without correction for multiple testing.
Results
Clinical characteristics
The study included samples from 264 individuals – 133 ADHD patients and 131 controls. Clinical characteristics of the participants, including smoking status based on cotinine levels, are listed in Table 1. As shown, male participants were underrepresented both in the patient (46.6%) and control (42.7%) groups, with a slightly and non-significantly lower proportion in the controls. The ADHD patients were older than the controls (mean age 28.7 compared to 22.7 years, P<0.001), and the age range was larger within the ADHD group (19–40 v. 18–33, data not shown). A lifetime presence of psychiatric disorders other than ADHD was frequently reported by the ADHD patients, with depression or anxiety being the most common (65.1%), whereas only a small number (n=7, 5.3%) of the controls responded that they had experienced any of the listed psychiatric disorders. As expected, the scores for current ADHD symptoms (ASRS score) were much higher among ADHD patients than controls, with mean scores of 46.1 and 22.9, respectively. Serum cotinine levels indicated that 66.1% of the ADHD patients were smokers, and 42% of these (27.8% of the total) were classified as heavy smokers. In comparison, only 12.3% of the controls were categorised as smokers (P<0.001). We received information on medical ADHD treatment for 123 of the patients, and it was reported that 92% of these had ever used central stimulant treatment and 80% (out of 114) still used it at the time of blood sample withdrawal (data not shown). For 19% of the patients, it was reported that antidepressants had been prescribed (lifetime prevalence).
Table 1 Clinical characteristics of the sample, including smoking status based on cotinine level analyses and P-values of group differences from chi-square and t-tests
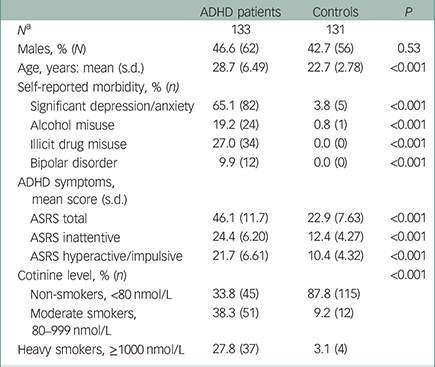
| ADHD patients | Controls | P | |
|---|---|---|---|
| N a | 133 | 131 | |
| Males, % (N) | 46.6 (62) | 42.7 (56) | 0.53 |
| Age, years: mean (s.d.) | 28.7 (6.49) | 22.7 (2.78) | <0.001 |
| Self-reported morbidity, % (n) | |||
| Significant depression/anxiety | 65.1 (82) | 3.8 (5) | <0.001 |
| Alcohol misuse | 19.2 (24) | 0.8 (1) | <0.001 |
| Illicit drug misuse | 27.0 (34) | 0.0 (0) | <0.001 |
| Bipolar disorder | 9.9 (12) | 0.0 (0) | <0.001 |
| ADHD symptoms, mean score (s.d.) | |||
| ASRS total | 46.1 (11.7) | 22.9 (7.63) | <0.001 |
| ASRS inattentive | 24.4 (6.20) | 12.4 (4.27) | <0.001 |
| ASRS hyperactive/impulsive | 21.7 (6.61) | 10.4 (4.32) | <0.001 |
| Cotinine level, % (n) | <0.001 | ||
| Non-smokers, <80 nmol/L | 33.8 (45) | 87.8 (115) | |
| Moderate smokers, 80–999 nmol/L | 38.3 (51) | 9.2 (12) | |
| Heavy smokers, ≥1000 nmol/L | 27.8 (37) | 3.1 (4) |
ADHD, attention-deficit hyperactivity disorder; ASRS, Adult ADHD Self-report Scale.
a The total number of participants in each analysis varies between 121 and 133 for the patients and 128 and 131 for the controls, because of missing items in the questionnaires.
Vitamin levels and ADHD status
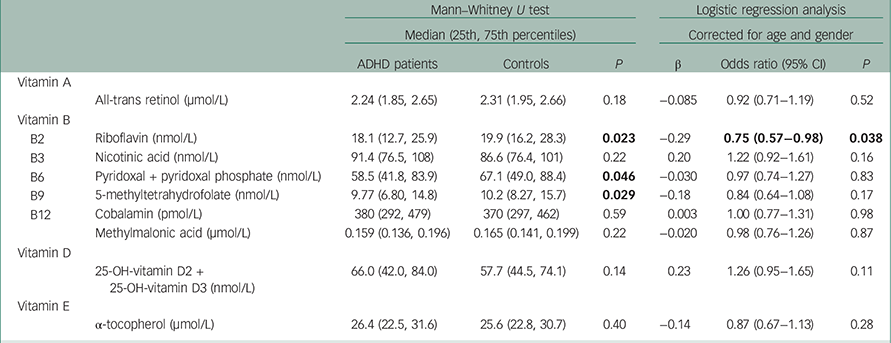
Table 2 shows the median (25th and 75th percentiles) serum vitamin concentrations for patients and controls. The concentrations of vitamins B2, B6 and B9 were all significantly lower in the ADHD group (P-values 0.023, 0.046 and 0.029, respectively). The table further shows the results from the logistic regression analyses with age and gender as covariates. Also in these analyses, lower levels of vitamin B2 showed a nominally significant association with ADHD (β=−0.29, P=0.038), whereas none of the other vitamins (including B6 and B9) was significantly associated with ADHD. Adjusting for current or previous alcohol abuse weakened the association with vitamin B2 (β=−0.27, P=0.060) but did not alter the other results (data not shown). Logistic regression analyses of the vitamins and ADHD status only including participants up to 30 years of age (n=205) also showed significant association between low levels of B2 and ADHD (β=−0.33, P=0.023), whereas no associations were seen for the other vitamins (data not shown).
Table 2 Serum vitamin levels in adult ADHD patients and controls
| Mann–Whitney U test | Logistic regression analysis | ||||||
|---|---|---|---|---|---|---|---|
| Median (25th, 75th percentiles) | Corrected for age and gender | ||||||
| ADHD patients | Controls | P | β | Odds ratio (95% CI) | P | ||
| Vitamin A | |||||||
| All-trans retinol (μmol/L) | 2.24 (1.85, 2.65) | 2.31 (1.95, 2.66) | 0.18 | −0.085 | 0.92 (0.71−1.19) | 0.52 | |
| Vitamin B | |||||||
| B2 | Riboflavin (nmol/L) | 18.1 (12.7, 25.9) | 19.9 (16.2, 28.3) | 0.023 | −0.29 | 0.75 (0.57−0.98) | 0.038 |
| B3 | Nicotinic acid (nmol/L) | 91.4 (76.5, 108) | 86.6 (76.4, 101) | 0.22 | 0.20 | 1.22 (0.92−1.61) | 0.16 |
| B6 | Pyridoxal + pyridoxal phosphate (nmol/L) | 58.5 (41.8, 83.9) | 67.1 (49.0, 88.4) | 0.046 | −0.030 | 0.97 (0.74−1.27) | 0.83 |
| B9 | 5-methyltetrahydrofolate (nmol/L) | 9.77 (6.80, 14.8) | 10.2 (8.27, 15.7) | 0.029 | −0.18 | 0.84 (0.64−1.08) | 0.17 |
| B12 | Cobalamin (pmol/L) | 380 (292, 479) | 370 (297, 462) | 0.59 | 0.003 | 1.00 (0.77−1.31) | 0.98 |
| Methylmalonic acid (μmol/L) | 0.159 (0.136, 0.196) | 0.165 (0.141, 0.199) | 0.22 | −0.020 | 0.98 (0.76−1.26) | 0.87 | |
| Vitamin D | |||||||
| 25-OH-vitamin D2 + 25-OH-vitamin D3 (nmol/L) | 66.0 (42.0, 84.0) | 57.7 (44.5, 74.1) | 0.14 | 0.23 | 1.26 (0.95−1.65) | 0.11 | |
| Vitamin E | |||||||
| α-tocopherol (μmol/L) | 26.4 (22.5, 31.6) | 25.6 (22.8, 30.7) | 0.40 | −0.14 | 0.87 (0.67−1.13) | 0.28 | |
Median, 25th and 75th percentiles, with P-values calculated from Mann–Whitney U tests and the results from logistic regression analyses corrected for age and gender.
Significance: P<0.05 is shown in bold.
Division of the vitamin levels into 10th percentiles revealed that for vitamins B2, B6 and B9, the patients were clearly overrepresented within the lowest bins, with 2.7-, 3.5- and 5.6-fold higher proportions of patients relative to controls in the lowest 10th percentile group, respectively (Fig. 1). The difference in the distribution between patients and controls was significant for vitamins B2 and B9 (P=0.017 and P=0.027, respectively), but not for vitamin B6 (P=0.13). The distribution of the vitamin levels was significantly different between the groups also for vitamin D (P=0.023). However, for vitamin D, ADHD patients were significantly overrepresented both in the lowest and highest 10th percentile groups (Fig. 1). The distributions were not significantly different between patients and controls for the other vitamins (data not shown).

Fig. 1 Distribution of the 10th percentiles of the levels of the vitamins B2, B6, B9 and D in ADHD patients and controls. P-values calculated from chi-squared tests. Significance: P<0.05.
To explore whether the individuals with levels in the lowest 10th percentile of vitamins B2, B6, B9 and D, and the highest of vitamin D, were different from the remaining participants, we compared their clinical characteristics with chi-square and t-tests, both in all participants together and in patients only. Generally, there were not significant differences between the participants with vitamin levels in the lowest 10th percentiles and the rest (data not shown). However, problems with illicit drugs were reported more frequently by participants with lower vitamin B6 levels (n=9; i.e. 47% of the patients in the lowest 10th percentile group compared to 24% of the remaining patients [P=0.034]). Furthermore, males were significantly overrepresented among the participants with vitamin D levels in the lowest 10th percentile (73% males, compared to 42% males in the remaining participants [P=0.002]), and being in the lowest vitamin D 10th percentile was associated with lower age when the patients were studied exclusively (P=0.037; mean age 25.7 v. 29.2 years). Having vitamin D levels in the highest 10th percentile was associated with female gender (81% females v. 52% females in the remaining participants [P=0.005]).
Vitamin levels and smoking
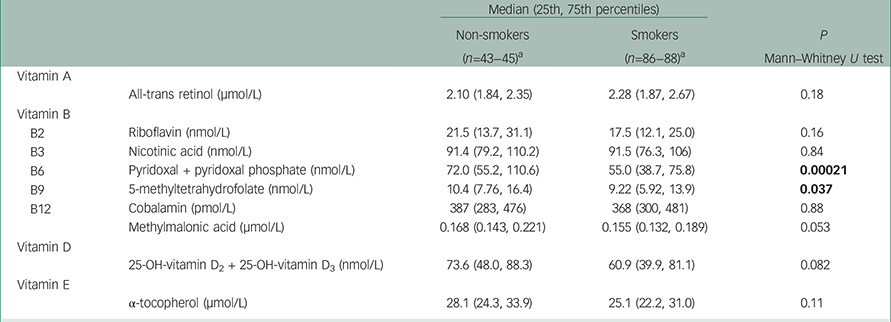
As shown in Table 3, vitamin B6 and B9 levels were significantly higher in non-smoking ADHD patients compared with smokers (P=0.00021 and P=0.037, respectively), whereas there were no significant differences for the other vitamins. The ANOVA analyses, where smokers were divided into moderate and heavy smokers, showed that the vitamin B6 levels were significantly different between the non-smokers and the moderate smokers (P=0.002) and also between non-smokers and heavy smokers (P=0.017) (data not shown). There was no difference between the two groups of smokers (P=0.905). The ANOVA analyses revealed no statistically significant group differences in vitamin concentration for the other vitamins, including vitamin B9 (P=0.065), where the largest difference was between non-smokers and heavy smokers (P=0.055).
Table 3 Median vitamin levels with 25th and 75th percentiles in non-smoking and smoking ADHD patients
| Median (25th, 75th percentiles) | ||||
|---|---|---|---|---|
| Non-smokers | Smokers | P | ||
| (n=43−45) a | (n=86−88) a | Mann–Whitney U test | ||
| Vitamin A | ||||
| All-trans retinol (μmol/L) | 2.10 (1.84, 2.35) | 2.28 (1.87, 2.67) | 0.18 | |
| Vitamin B | ||||
| B2 | Riboflavin (nmol/L) | 21.5 (13.7, 31.1) | 17.5 (12.1, 25.0) | 0.16 |
| B3 | Nicotinic acid (nmol/L) | 91.4 (79.2, 110.2) | 91.5 (76.3, 106) | 0.84 |
| B6 | Pyridoxal + pyridoxal phosphate (nmol/L) | 72.0 (55.2, 110.6) | 55.0 (38.7, 75.8) | 0.00021 |
| B9 | 5-methyltetrahydrofolate (nmol/L) | 10.4 (7.76, 16.4) | 9.22 (5.92, 13.9) | 0.037 |
| B12 | Cobalamin (pmol/L) | 387 (283, 476) | 368 (300, 481) | 0.88 |
| Methylmalonic acid (μmol/L) | 0.168 (0.143, 0.221) | 0.155 (0.132, 0.189) | 0.053 | |
| Vitamin D | ||||
| 25-OH-vitamin D2 + 25-OH-vitamin D3 (nmol/L) | 73.6 (48.0, 88.3) | 60.9 (39.9, 81.1) | 0.082 | |
| Vitamin E | ||||
| α-tocopherol (μmol/L) | 28.1 (24.3, 33.9) | 25.1 (22.2, 31.0) | 0.11 | |
a The number of participants varies according to how many had the level of each vitamin successfully analysed.
Significance: P<0.05 is shown in bold.
Vitamin levels and ADHD symptoms
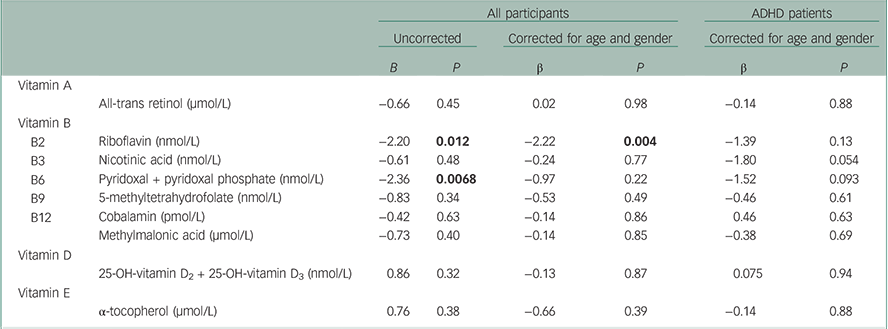
As shown in Table 4, we performed linear regression analyses of the relationship between vitamin levels and ASRS scores in all participants together, both with and without correction for age and gender. The uncorrected analyses showed significant association between higher scores on the ASRS and lower levels of vitamins B2 and B6 (β=−2.20 and P=0.012, and β=−2.36 and P=0.0068, respectively), whereas only vitamin B2 was significantly associated after correction for age and gender (β=−2.22 and P=0.004). Because the mean ASRS scores were significantly lower in the controls (see Table 1), the analyses were also performed separately in the ADHD patients, with correction for age and gender. No significant associations were observed from these analyses. Adjusting for age and gender, an additional analysis was also performed in participants who were 30 years or younger. Also in this group of patients, there was a trend associating higher ASRS symptom scores with lower levels of vitamin B2 (β=−1.89 and P=0.189, n=69). This effect was stronger when patients and controls were analysed together (β=−2.63 and P=0.005, n=193). In the combined group, the effect was significant, possibly because of the increased statistical power and the added variance in symptom scores when all study participants were analysed together. We also performed analyses of a putative effect of smoking by adding cotinine levels (grouped as non-, medium and heavy smoking) as a covariate, together with age and gender. Again, a significant association was observed only for vitamin B2, where lower levels were associated with higher ASRS scores in all participants together (β=−1.97 and P=0.007), but not in the patients alone (β=−1.38 and P=0.133) (data not shown).
Table 4 Linear regression analyses of total ADHD symptom scores on the ASRS and serum vitamin levels, in all participants together (119–123 ADHD patients and 126–127 controls) and the ADHD patients separately
| All participants | ADHD patients | ||||||
|---|---|---|---|---|---|---|---|
| Uncorrected | Corrected for age and gender | Corrected for age and gender | |||||
| B | P | β | P | β | P | ||
| Vitamin A | |||||||
| All-trans retinol (μmol/L) | −0.66 | 0.45 | 0.02 | 0.98 | −0.14 | 0.88 | |
| Vitamin B | |||||||
| B2 | Riboflavin (nmol/L) | −2.20 | 0.012 | −2.22 | 0.004 | −1.39 | 0.13 |
| B3 | Nicotinic acid (nmol/L) | −0.61 | 0.48 | −0.24 | 0.77 | −1.80 | 0.054 |
| B6 | Pyridoxal + pyridoxal phosphate (nmol/L) | −2.36 | 0.0068 | −0.97 | 0.22 | −1.52 | 0.093 |
| B9 | 5-methyltetrahydrofolate (nmol/L) | −0.83 | 0.34 | −0.53 | 0.49 | −0.46 | 0.61 |
| B12 | Cobalamin (pmol/L) | −0.42 | 0.63 | −0.14 | 0.86 | 0.46 | 0.63 |
| Methylmalonic acid (μmol/L) | −0.73 | 0.40 | −0.14 | 0.85 | −0.38 | 0.69 | |
| Vitamin D | |||||||
| 25-OH-vitamin D2 + 25-OH-vitamin D3 (nmol/L) | 0.86 | 0.32 | −0.13 | 0.87 | 0.075 | 0.94 | |
| Vitamin E | |||||||
| α-tocopherol (μmol/L) | 0.76 | 0.38 | −0.66 | 0.39 | −0.14 | 0.88 | |
ADHD, attention-deficit hyperactivity disorder; ASRS, Adult ADHD Self-report Scale.
Significance: P<0.05 is shown in bold.
Discussion
As vitamins are essential nutrients involved in brain development and neurological functions, Reference Bender4 it may be hypothesised that vitamin deficiency could contribute to ADHD symptoms. It has recently been reported that serum levels of vitamin D are lower in children with ADHD compared with healthy controls. Reference Bener, Kamal, Bener and Bhugra5–Reference Kamal, Bener and Ehlayel7 In a study comparing oxidative stress markers in adult ADHD patients and controls, vitamin B9 (folate) was higher in the patients, whereas there was no difference between the groups for vitamin B12. Reference Karababa, Savas, Selek, Cicek, Cicek and Asoglu10 Two randomised placebo-controlled trials from 2014 support the theory of a relationship between low vitamin levels and ADHD symptoms; Rucklidge et al reported that symptom severity was significantly reduced in adult ADHD patients who received food supplements containing different micronutrients including vitamins and that this was accompanied by a significant increase in serum vitamin levels. Reference Rucklidge, Frampton, Gorman and Boggis8 Manor and co-workers found that symptoms in adult ADHD patients were significantly reduced after administration of the vitamin B6-related drug metadoxine. Reference Manor, Rubin, Daniely and Adler9
For most of the vitamins, we observed no consistent association between vitamin levels and ADHD diagnosis or symptoms. However, lower levels of vitamins B2, B6 and B9 were significantly associated with ADHD, and lower vitamin B2 and B6 levels were associated with higher ADHD symptoms scores. There were also significant differences in the distribution of vitamin D levels between the groups, with the lowest and highest levels being overrepresented in the patients.
Lower levels of B2 were also associated with increased ASRS scores, even when adjusting for smoking, when patients and controls were analysed together. For vitamin B6, lower levels were also associated with a diagnosis of ADHD. However, lower levels of vitamin B6 were strongly associated with smoking (P=0.00021), which was more common in the patients, and previous or current problems with illicit drugs were also significantly more common among the patients with vitamin B6 levels in the lowest 10th percentile. As opposed to the results from Karababa and co-workers, Reference Karababa, Savas, Selek, Cicek, Cicek and Asoglu10 the median level of vitamin B9 was significantly lower in ADHD patients than in controls. However, lower vitamin B9 levels were also associated with smoking, and the results from the logistic regression, as well as the analyses of ASRS, did not show significant association with this vitamin. The distribution of vitamin D was different in patients and controls, with the patients being overrepresented both in the lowest and highest 10th percentiles. Examination of clinical characteristics showed that having vitamin D levels of the lowest 10th percentile was associated with male gender, whereas females were more abundant among the individuals with the highest vitamin D levels.
It was recently reported that the nutritional status and intake of vitamins was significantly lower in children with ADHD compared with healthy controls. Reference Dura-Trave and Gallinas-Victoriano25 The authors suggested that pharmacological treatment with methylphenidate could play a role in the lower vitamin intake, possibly through alterations in eating preferences or appetite. Reference Dura-Trave and Gallinas-Victoriano25 A potential influence of stimulant treatment could be of relevance for our results, because, as described in our previous publication, almost all of the ADHD patients from whom we had this information had tried central stimulant treatment (92%, n=123), at least previously. Reference Aarsland, Landaas, Hegvik, Ulvik, Halmoy and Ueland26 Dietary habits acquired in childhood often persist through to adulthood, Reference Kelder, Perry, Klepp and Lytle27 and inadequate vitamin intake in children with ADHD could suggest that the vitamin intake may also be poorer in adult ADHD patients than in controls.
Vitamins are consumed as part of the ordinary diet and as vitamin supplements. Vitamin D in particular has been subject to much attention, and vitamin supplements have been suggested to be beneficial for a number of complaints. One reason for the overabundance of ADHD patients in the highest 10th percentiles of vitamin D may thus be that relatively more ADHD patients take vitamin D supplements, either as part of an experimental treatment of symptoms or as a consequence of a diagnosed vitamin D deficiency. This could explain our observation that patients were overrepresented both in the highest and lowest 10th percentile, possibly representing both patients who take supplements and those who have an untreated vitamin D deficiency, respectively. Although this is hypothetical, these findings could be in agreement with the results from the studies in children and adolescents, where the vitamin D levels were lowest in patients. Reference Bener, Kamal, Bener and Bhugra5–Reference Kamal, Bener and Ehlayel7 Interestingly, females were overrepresented among the ADHD patients with high levels of vitamin D, whereas males had low levels of vitamin D, possibly consistent with a higher health-seeking behaviour or more exposure to ultraviolet radiation of the skin in females.
If the low vitamin levels in ADHD are caused by differences in uptake or metabolism because of genetic factors, the lower levels in adult ADHD patients might reflect a lower level also during childhood. The effect on ADHD, which is considered a neurodevelopmental disorder, might thus be that the lower vitamin levels have influenced brain development at an earlier age. Regardless of whether that is the case, lower vitamin levels may also influence the ADHD symptoms in adulthood directly, as indicated by the reports of reduced symptoms in adult ADHD patients after the intake of food supplements with vitamins. Reference Rucklidge, Frampton, Gorman and Boggis8,Reference Manor, Rubin, Daniely and Adler9 Vitamins act as important cofactors in various biochemical pathways in the brain, and especially B vitamins play important roles in the formation of different neurotransmitters, such as dopamine and serotonin, which are involved in ADHD-related traits and are affected by treatment with central stimulants. Reference Rucklidge, Frampton, Gorman and Boggis8
Smoking and alcohol
As smoking frequently co-occurs with ADHD, Reference Nigg28 it is difficult to recruit representative groups of patients and controls without having different proportions of smokers. This is problematic, as vitamin levels have been found to vary with smoking status. Reference Yanbaeva, Dentener, Creutzberg, Wesseling and Wouters29,Reference Walmsley, Bates, Prentice and Cole30 Smokers and non-smokers have different dietary habits, including a lower intake of certain vitamins in smokers. Reference Cade and Margetts31,Reference Subar, Harlan and Mattson32 Additionally, smokers seem to have lower levels of several vitamins even when controlling for diet, possibly because of oxidative stress caused by smoking. Reference Yanbaeva, Dentener, Creutzberg, Wesseling and Wouters29,Reference Ulvik, Ebbing, Hustad, Midttun, Nygard and Vollset33 To examine current smoking status of our participants, we measured plasma levels of cotinine. Cotinine is a nicotine metabolite with a half-life of 14–20 h (in contrast to nicotine's 2 h) and is a commonly used biomarker for nicotine intake. To distinguish smokers from non-smokers, we chose the widely used cut-off of 80 nmol/L. Reference Jarvis, Tunstall-Pedoe, Feyerabend, Vesey and Saloojee24 Using this definition, 66% of the ADHD patients were categorised as smokers. In the control group, the proportion of smokers was significantly lower (12%). The number of smokers in our control group is comparable to Statistics Norway's data (www.ssb.no), which show that the percentages of daily smokers in the age group 16–24 years were 23% and 12% in the years 2005 and 2010, respectively.
Because smoking status is closely related to the ADHD diagnosis, including it as a covariate in the analyses would possibly attenuate associations with ADHD. To examine the possible effect of smoking on vitamin concentrations, we instead compared their median levels in ADHD patients defined as non-smokers and smokers. We did not perform the analyses in the control group, as it contained too few smokers to obtain reliable results. These analyses showed significant association between smoking status and lower levels of vitamins B6 and B9, which is in line with previous reports. Reference Ulvik, Ebbing, Hustad, Midttun, Nygard and Vollset33 We thus cannot rule out that the observed associations between these vitamins and ADHD are mediated by differences in smoking status between the groups.
A high consumption of alcohol over time is known to influence vitamin levels and cause vitamin B deficiency. Among the ADHD patients, 19.2% reported present or past problems with alcohol use. In comparison, only 0.8% (one individual) of the controls reported such problems. Bearing in mind that this number was very different between the groups, we also performed the logistic regression analyses of vitamin levels and ADHD status with problems with alcohol abuse added as a covariate. The association with vitamin B2 was weakened by this analysis (from β=−0.29 and P=0.038 to β=−0.27 and P=0.060), but the adjustment did not otherwise influence the results.
Strengths and limitations of the sample
An advantage with our sample is that it is quite large, as it includes 133 patients and 131 controls. This is considerably larger than most previous studies in ADHD, including the vitamin studies by the groups of Goksugur, Rucklidge and Karababa, Reference Goksugur, Tufan, Semiz, Gunes, Bekdas and Tosun6,Reference Rucklidge, Frampton, Gorman and Boggis8,Reference Karababa, Savas, Selek, Cicek, Cicek and Asoglu10 and probably sufficiently large to detect major differences in vitamin levels between the groups. Still, replication of our results in other and preferably larger samples is warranted in order to further evaluate the impact of vitamin levels in ADHD.
It is a further strength of our study that gender was rather similarly distributed in the patient and control groups and that both genders were strongly and almost equally represented, as opposed to the majority of ADHD studies, where most of the participants were male. However, the unequal age distribution between the groups is a limitation of our study, as the mean age was significantly higher in the patient group (28.7 v. 22.7 years, P<0.001). We are not aware of any age-dependent changes in vitamin levels in the age range from 18 to 40 years, but cannot exclude the possibility. Therefore, age was included as a covariate in the analyses, and additional analyses including only participants up to 30 years of age were performed.
In particular for vitamin D, it is known that blood levels vary during the year, as a result of exposure to sunlight and dietary habits. Reference Loken-Amsrud, Holmoy, Bakke, Beiske, Bjerve and Bjornara34 In both ADHD patients and controls, blood samples were collected throughout the year. Most samples from patients were collected in October and November, and fewest in May and December, whereas the majority of control samples were collected from November to February. In a study of Norwegian multiple sclerosis patients, it was found that vitamin D levels were highest in August and September and lowest in March and April. Reference Loken-Amsrud, Holmoy, Bakke, Beiske, Bjerve and Bjornara34 Based on these data, we don't consider that the levels of vitamin D or the other data reported here were systematically biased by the time of sample collection.
The majority of blood samples were obtained from their GPs for the patients and at the project laboratory facilities in Bergen for the majority of controls, which resulted in a group difference in the transportation and handling of the samples. However, all metabolites included in this study were considered to be sufficiently stable during the estimated time required for transport and analysis.
The reason why we observed association between lower levels of some vitamins and ADHD is uncertain and probably multifactorial. Regrettably, we do not have information on lifestyle and nutrient intake from the participants to help in the interpretation of our observations. It is reasonable to think that differences in dietary factors may partly be responsible for the differences.
The P-values reported here were not adjusted for multiple testing. If applying Bonferroni correction for all P-values listed in Tables 2, 3 and 4 (n=54), the threshold for significance would be 0.05/54=0.000925. Using this correction, there was still a significant difference in vitamin B6 levels between smokers and non-smokers (Table 3) (P=0.00021). With false discovery rate correction for a total of 54 comparisons, the difference in vitamin B6 levels between non-smoking and smoking ADHD patients remained significant (P=0.011), whereas none of the other P-values were significant. However, the levels of the metabolites included in this study are not independent of each other, and applying such corrections is probably too conservative.
Suggestions for further research
Future studies examining nutrient levels should ideally be performed in larger samples, with sufficient statistical power to detect smaller group differences. This would allow comparisons between clinical subgroups, at different ages and across different symptom dimensions and comorbid conditions. The recruitment of clinically representative patient samples and controls that are matched for age, sex and socio-economic status is important. Information about lifestyle parameters, including reliable information regarding past and present dietary habits will aid the interpretation of the results and whether ADHD patients have altered metabolism of nutrients. Pre- and postnatal exposures to nutrients, substances of abuse and medication should also be considered. Access to additional biomarkers in biochemical pathways that are dependent on the respective vitamins can show the functional consequences of vitamin deficiency. Likewise, molecular genetic data can reveal differences in vitamin metabolism, also in ethnic groups and in clinical subgroups. Ideally, studies should be conducted across cultures with different dietary traditions, ethnicities and geographical areas.
We have investigated eight different vitamins, making this the most extensive study of vitamins in ADHD so far and also the first study of many of the vitamins in relation to ADHD. Lower levels of the vitamins B2, B6 and B9 were associated with the ADHD diagnosis, and lower levels of B2 and B6 were also associated with a higher symptom load. However, lower vitamin B6 and B9 levels were associated with smoking, which may have influenced the results. The lowest and highest values of vitamin D were overrepresented in ADHD patients, indicating a higher prevalence of vitamin D deficiency, possibly also treated. We have, however, no clinical information on supplement treatment, and group differences in the time of collection of blood samples may have influenced our results.
It is possible that low levels of certain vitamins may contribute to ADHD symptoms. Dietary intervention trials have shown promising effects in ADHD. Thus, identification and correction of low vitamin levels could be beneficial in treatment of ADHD. Further studies are warranted for replication and for examination of the underlying mechanisms.
Acknowledgements
We thank all patients and controls who volunteered to participate in this study, and Lisa Vårdal, Johanne Telnes Instanes and Tor-Arne Hegvik for their work with patient recruitment and data collection.
Funding
This work was supported by Stiftelsen K.G. Jebsen, University of Bergen, Western Norway Health Authorities (Helse Vest), European Community's Seventh Framework Programme under grant agreement number 602805 and the H2020 Research and Innovation Program under grant agreement numbers 643051 and 667302.











eLetters
No eLetters have been published for this article.